 Samples with IHC score of 2+ are considered as inconclusive and a second evaluation with fluorescent in situ hybridization (FISH) methodology is required. Females with an IHC score 3+ are candidates for the costly herceptin treatment. Immunohistochemistry (IHC) is the first methodology selected for Her-2 protein evaluation since it is the most economical however, it is not the most precise. The discovery of her-2 gene overexpression in BC tissues and its association with aggressive clinical behavior has generated diagnostic interest by using her-2 gene expression as a predictive marker for herceptin treatment response ( 1– 3). Clinical studies have shown that her-2 gene upregulation and Her-2 protein overproduction predicts poor prognosis in BC patients lacking estrogen and progesterone receptors, inducing a high rate of cell proliferation and tumor chemotherapy resistance ( 2, 3). High levels of her-2 have also been found in several ovarian and endometrial tumors ( 1). her-2 is one of the most frequently studied BC genes due to its overexpression observed in 20–30% of aggressive cases. In human cancer, oncogenes such as her-2 have been identified and used as molecular markers. Lately, the number of BC fatalities has increased approximately to 40,000 annually in Mexico ( ). Tumor factors and pCR outcomes of patients 2.Breast cancer (BC) is one of the most common causes of mortality among females, accounting for 23% of total cancer cases and 14% of cancer mortality around the world (WHO, 2010). The rates of pCR were 29.3% in the AR-positive group and 43.2% in the AR-negative group (p=0.007). There was a significant difference between the AR-positive group and AR-negative group regarding subtypes (p < 0.001). In the AR-negative group, the HR−/HER2− subtype was most frequent (72.6%), followed by the HR+/HER2− subtype (14.7%). Regarding the subtypes, HR+/HER2−, HR+/HER2+, HR−/HER2+, and HR−/HER2− subtypes comprised 46.9%, 23.6%, 20.4%, and 9.1% of the AR-positive group, respectively. Ki-67 levels were significantly lower in the AR-positive group in comparison to the AR-negative group (38.6☒0.1 vs. The AR-positive group had significantly higher ER positivity and PR positivity, and HER2 positivity than the AR-negative group. Regarding the AR receptor status, the AR-positive group had significantly higher clinical T stages 3, and 4 than the AR-negative group (32.3% vs. The mean age (±standard deviation) at diagnosis was 50.0☙.6 years. Five hundred twenty-nine patients (84.8%) were AR-positive and 95 patients (15.2%) were AR-negative. Table 1 summarized the clinicopathologic characteristics of the patients. Characteristics of the study populationĪ total of 624 breast cancer patients who underwent neoadjuvant chemotherapy were included in the analysis. Characteristics of the study population 1. 4.1.2 (R Core Team, Vienna, Austria), and a p-value < 0.05 was considered statistically significant. All statistical analyzes were performed using R ver. In addition, after correcting for significant factors, to confirm the effect of pretreatment AR on pCR, the distribution of pretreatment AR and pCR in subgroups by subtype was summarized using percentages, and the relationship was confirmed using the chi-square test or Fisher exact test. 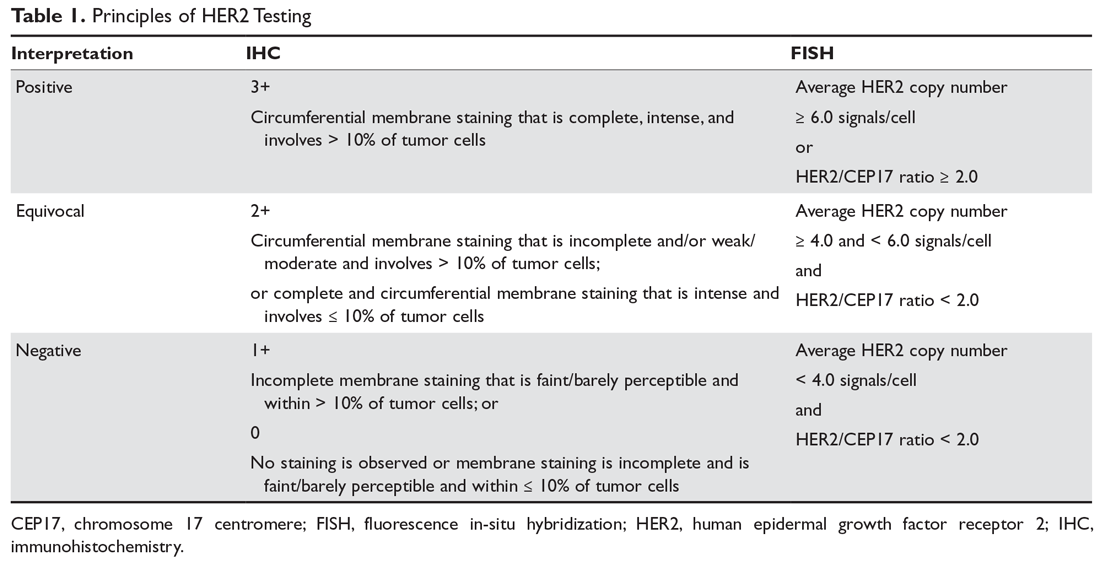
After including all clinical factors, the backward elimination method with criterion p-value < 0.05 was used to identify significant factors. Clinical factors affecting pCR were identified using a multivariable logistic regression model. For clinical factors according to pretreatment AR and pCR, depending on the types of variables, the chi-square test or Fisher exact test was used for categorical variables, and the t test was used for continuous variables. Patient characteristics were summarized by the mean and standard deviation for continuous variables, and frequency counts with percentages for categorical variables. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
